About our group
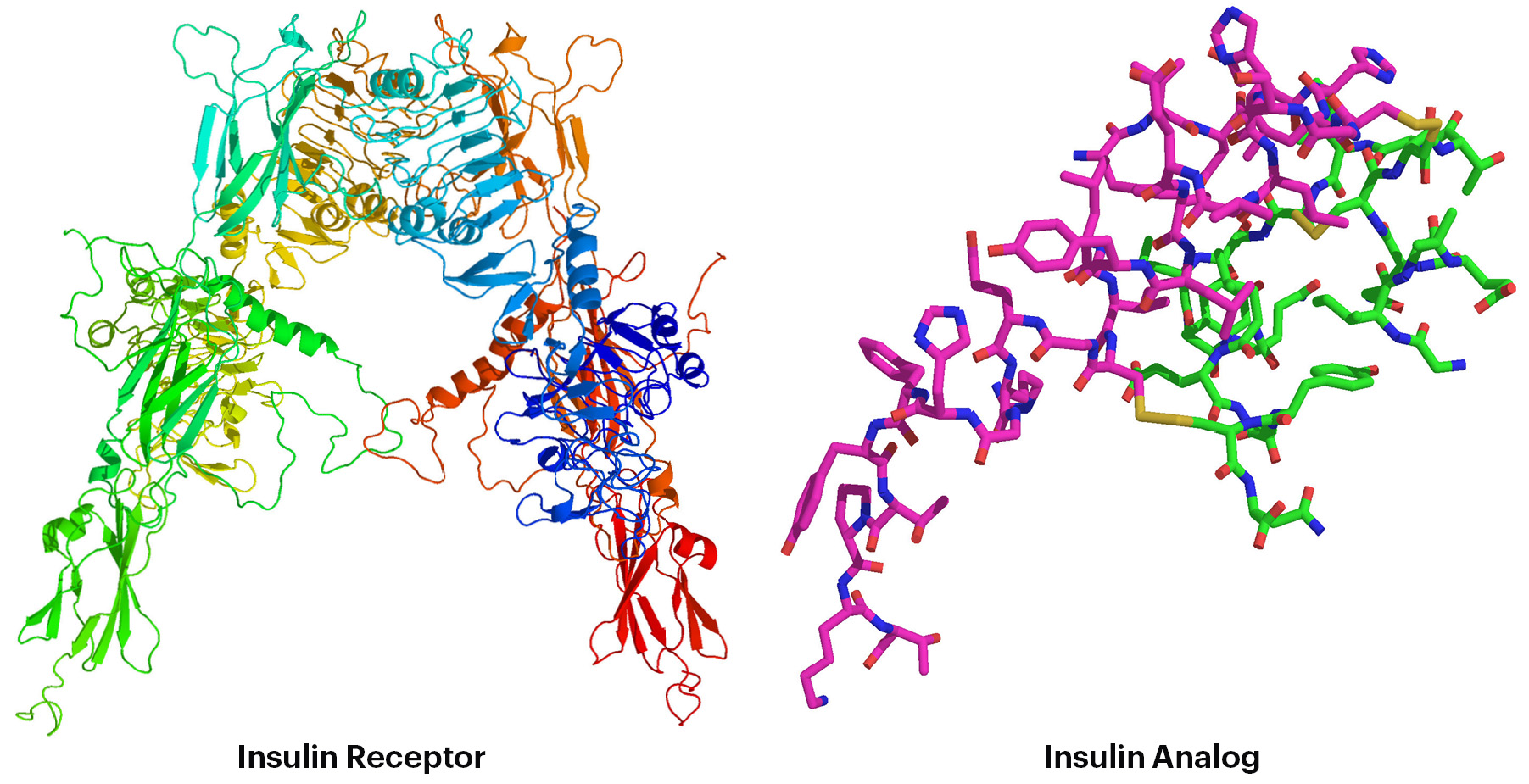
On the basis of our long lasting experience in the quantum chemical description of noncovalent interactions in biomacromolecules, we have developed quantum mechanics-based scoring function and have applied it in virtual screening. We have successfully employed our methodology to series of inhibitors of cyclin-dependent kinases, HIV-1 protease, aldose reductase and parasitic cysteine peptidase. Currently, we focus on insulin analogs and mimetics using the structures of the insulin receptor. The generality of the newly developed quantum mechanics-based scoring functions has been tested on an extended dataset of diverse protein-ligand complexes and their performance was compared to that of widely used scoring functions from academia and industry. We have found that our scoring functions clearly outperform the standard functions. We propose this method for general use in computer-aided drug design. Our new project concerns the application of noncovalent interactions in materials science. Electronic and spin states of metal-containing organic molecules can be tuned by their noncovalent interactions with metallic and carbon-containing surfaces.