About our group
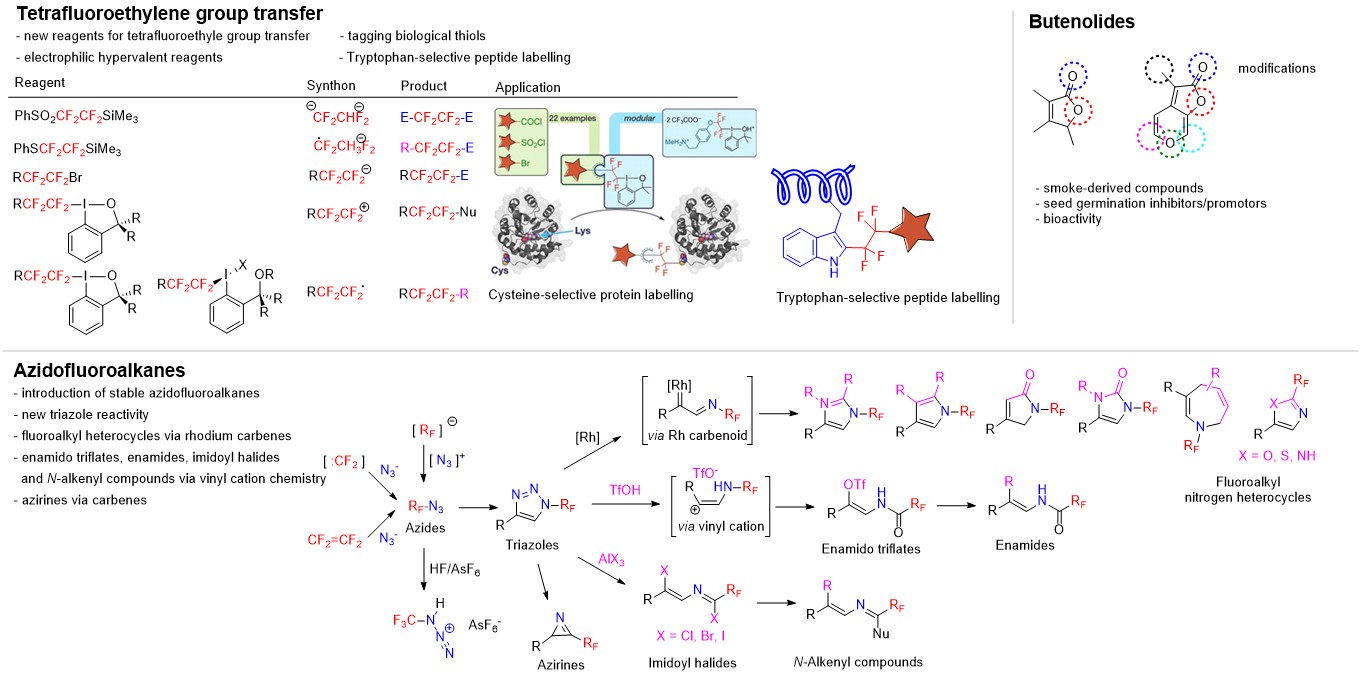
The central theme of our research is in the development of new, selective and convenient synthetic reagents and methods towards novel organic molecules, which can find applications in crop protection, drug design and in materials. We study new reactions and their mechanisms. We are focusing on organic chemistry of the main group elements such as fluorine, phosphorus, silicon, sulfur and iodine. We have been active in methodology development of fluorinated phosphonates, pentafluorosulfanyl (SF5) containing compounds, fluoroalkylations, transfer of tetrafluoroethyl and tetrafluoroethylene fragments, bioconjugations, and the chemistry of fluorinated azides, heterocycles, enamines and enamides.