Prof. Cynthia J. Burrows (University of Utah, Salt Lake City, UT, USA)
Mechanisms of Guanine Oxidation Leading to DNA Mutations
Abstract
Oxidative stress is thought to underlie many disorders from cancer and aging to neurological and heart diseases. DNA bases, particularly guanine, are a key target of reactive oxygen species and transition metal-mediated oxidation, leading to a wealth of heterocyclic chemistry, and unfortunately, a range to mutations in the genome.
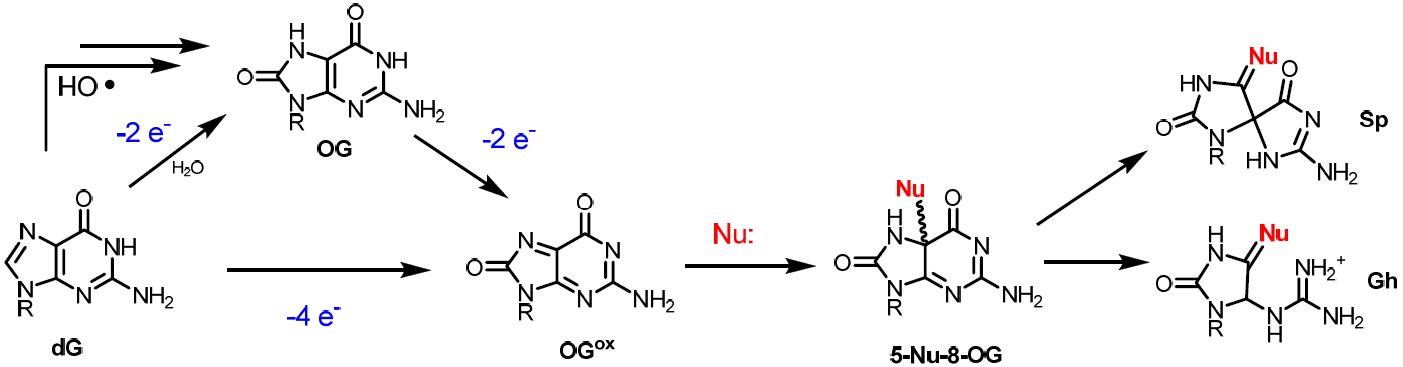
The common DNA oxidation product 8-oxoguanosine is highly sensitive to further oxidation generating a family of hydantoin lesions including spiroiminodihydantoin (Sp) and guanindinohydantoin (Gh). In the presence of nucleophiles besides water, analogous adducts are formed in which one of the oxo moieties of Sp or Gh is replaced by nucleophiles such as spermine and lysine. Both the reactivity and the product distribution of 8-oxoG oxidation are influenced by the local context of the lesion. In addition, the ability of DNA repair enzymes such as hNEIL1 to recognize and excise the Sp and Gh substrates is dependent on the base opposite the lesion as well as the strand structure. The study of organic reaction mechanisms lends insight both toward the generation of DNA damage and the ability of enzymes to process the damage sites.