Prof. Gregory C. Fu (Massachusetts Institute of Technology, Cambridge, MA, USA)
Palladium- and Nickel-Catalyzed Coupling Reactions of Alkyl Electrophiles
Abstract
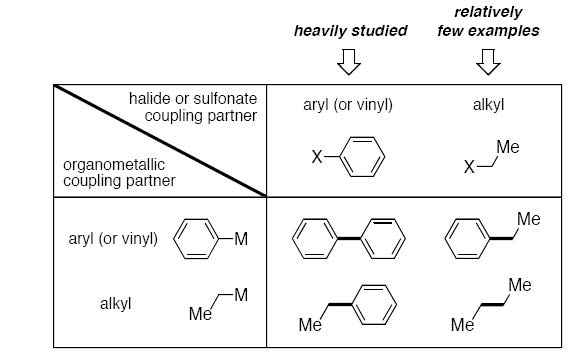
Despite the tremendous accomplishments that have been described in the development of palladium- and nickel-catalyzed carbon–carbon bond-forming processes, it is nevertheless true that many significant opportunities remain. For example, to date the overwhelming majority of studies have focused on couplings between two sp2-hybridized reaction sites (e.g., an aryl metal with an aryl halide).
As of 2001, there were few examples of palladium- or nickel-catalyzed coupling reactions of alkyl electrophiles. During the past several years, we have pursued the discovery of palladiumand nickel-based catalysts for coupling activated and unactivated primary and secondary alkyl electrophiles that bear β hydrogens. Our recent efforts to develop broadly applicable methods, including enantioselective processes, will be discussed.