
Prof. Alois Fürstner (Max-Planck-Institut für Kohlenforschung, Mülheim, Germany)
Catalysis for Total Synthesis
Abstract
This lecture will provide an up-date on our ongoing programs concerning the total synthesis and evaluation of complex natural products of biological significance. Targets of current interest include the latrunculin family of actin binding macrolides [1], the highly cytotoxic polyene iejimalide [2], various members of the amphidinolide series [3-5], complex glycolipids such as ipomoeassin [6], and the telomerase inhibitors dictyodendrin A-E [7]. All syntheses are largely catalysis-based, featuring the scope of methodology under scrutiny in this laboratory (ring closing alkene- and alkyne metathesis, iron catalyzed coupling reactions, titanium-induced heterocycle syntheses).
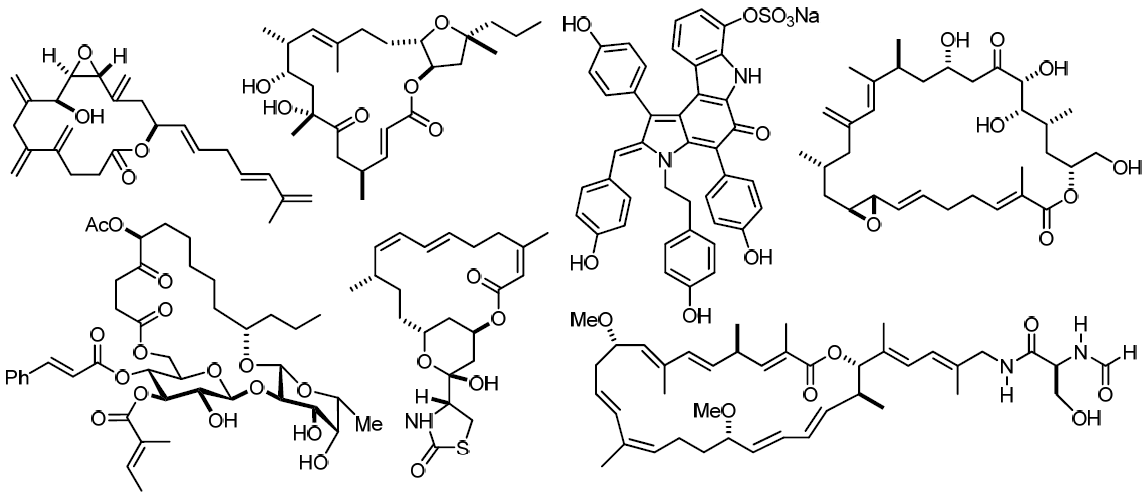
References
- A. Fürstner, D. Kirk, M. D. B. Fenster, C. Aissa, D. De Souza, O. Müller Proc. Natl. Acad. Sci. USA 2005,
102, 8103.
- A. Fürstner, C. Nevado, M. Waser, M. Tremblay, C. Chevrier, F. Teply, C. Aïssa, E. Moulin, O. Müller J. Am. Chem. Soc. 2007, 129, 9150.
- A. Fürstner, E. Kattnig, O. Lepage J. Am. Chem. Soc. 2006, 128, 9194.
- A. Fürstner, O. Larionov, S. Flügge Angew. Chem. Int. Ed. 2007, 46, 5545.
- A. Fürstner, L. C. Bouchez, J.-A. Funel, V. Liepins, F.-H. Porée, R. Gilmour, F. Beaufils, D. Laurich, M. Tamiya Angew. Chem. Int. Ed. 2007, 46, 9265.
- A. Fürstner, T. Nagano J. Am. Chem. Soc. 2007, 129, 1906.
- A. Fürstner, M. M. Domostoj, B. Scheiper J. Am. Chem. Soc. 2006, 128, 8087.





