
Prof. E. Peter Kündig (University of Geneva, Switzerland)
Advances in Transition Metal Lewis Acid – Mediated and Catalyzed Reactions
Abstract
New powerful applications in asymmetric synthesis and catalysis have put half-sandwich complexes of the type MCpLn and M(arene)Ln into the limelight. Efficient synthetic access and new ligands are instrumental to further development and new applications. This lecture will focus on several aspects of the synthesis of electron-poor half sandwich complexes.
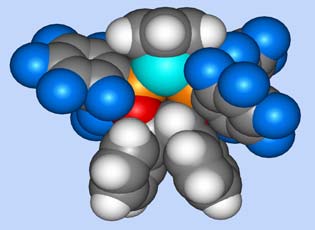
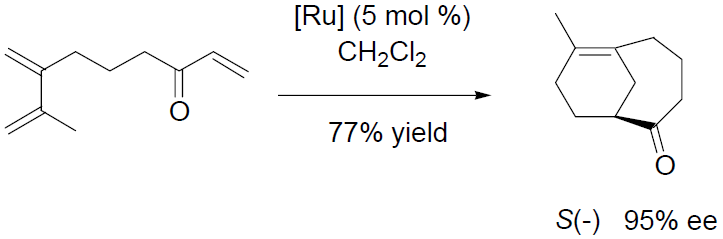
The 16-electron half-sandwich complexes CpM(P*P)]+ (M = Fe, Ru) and [IndRu(P*P)]+ where P*P are electron poor bidentate fluoroaryl-phosphinite ligands are efficient catalysts for asymmetric [4+2] cycloaddition reactions between enals and dienes, and for the [3+2] cycloaddition reactions between enals and nitrones. Attractive features of this family of Lewis acid catalysts are their straightforward syntheses, well-defined structures and tunable electronic properties.1 The high O stability of the Ru complexes allows quantitative catalyst recovery. New developments include inter- and intramolecular [4+2] cycloadditions involving α,β-unsaturated ketones.2
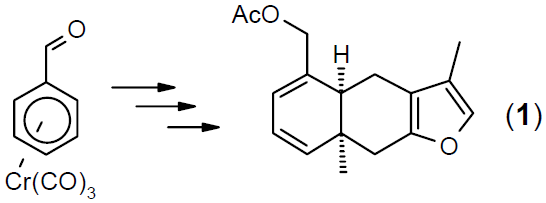
Temporary complexation of an arene by the electrophilic Cr(CO)3 group is the key for the transformation of arenes into substituted alicyclic molecules.3 A number of highly asymmetric variants have been developed and have been applied to the synthesis of natural products or fused ring systems of synthetic interest.4 An example is the synthesis of both enantiomers of acetoxytubipofuran (1).4a Complexation of an arene to an electrophilic metal fragment also provides new opportunities for the enantioselective catalytic desymmetrization of meso-complexes.5
References
- Alezra, V.; Bernardinelli, G.; Corminboeuf, C.; Frey, U.; Kündig, E. P.; Merbach, A. E.; Saudan, C. M.; Viton, F.; Weber, J. J. Am. Chem. Soc. 2004, 126, 4843-4853 and ref. cit..
- Rickerby, J.; Vallet, M.; Bernardinelli, G.; Viton, F.; Kündig, E. P. Chem. Eur. J. 2007, ASAP.
- Kündig, E. P., Transition Metal Arene p-Complexes in Organic Synthesis and Catalysis. Springer Verlag: Heidelberg, 2004; Vol. 7.
- a) Kündig, E. P.; Laxmisha, M. S.; Cannas, R.; Tchertchian, S.; Liu, R. G. Helv. Chim. Acta 2005, 88, 1063-1080. b) Kündig, E. P.; Bellido, A.; Kaliappan, K. P.; Pape, A. R.; Radix, S. Org. Biomol. Chem. 2006, 4, 342-351.
- c) Kündig, E. P.; Enriquez Garcia, A.; Lomberget, T.; Bernardinelli, G. T. Angew. Chem. Int. Ed. Engl. 2006, 45, 98-101. d) Kündig, E. P.; Chaudhuri, P. D.; House, D.; Bernardinelli, G. T. Angew. Chem. Int. Ed. Engl. 2006, 45, 1092-1095. e) Cumming, R. G.; Bernardinelli, G.; Kündig, E. P., Chem. Asian J. 2006, 1, 453-458.








