Prof. Julius Rebek, Jr. (Scripps Research Institute, La Jolla, CA, USA)
Molecular Assembly and Encapsulation
Abstract
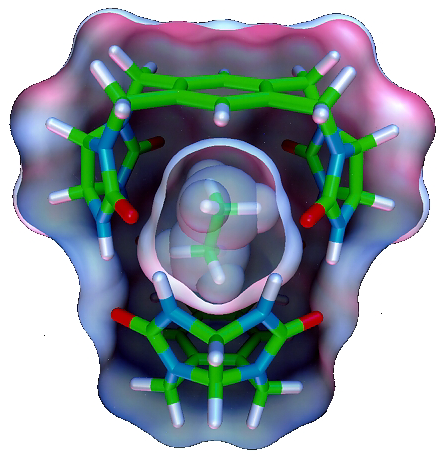
Reversible encapsulation creates spaces where molecules are temporarily isolated from others in solution. Molecules are held within the space of the capsule for lifetimes ranging from milliseconds to hours, and conventional NMR spectroscopy is used to report on the chemical and magnetic environment as well as the arrangement of molecules in the encapsulation complex. The complexes self-assemble when, and only when, the spaces inside the capsules are appropriately filled. The weak intermolecular forces that hold these self-assemblies together allow equilibration of the encapsulation complexes at ambient temperatures and pressures and in the liquid phase. When two or more molecules are encapsulated, intermolecular phenomena are revealed that cannot be observed by other methods. We describe here the unique behavior that emerges from reversible encapsulation, including new forms of stereochemistry, isomerism, asymmetry and reactivity of molecules held at close range. Questions such as: What’s it like inside? What are the rules that govern the inner space? Does catalysis occur inside? Can the space be made chiral? will be addressed in the context of reversible encapsulation complexes. A rendering of ethane inside a small capsule is shown below.