
Prof. Antonio M. Echavarren (Institute of Chemical Research of Catalonia, Tarragona, Spain)
Gold Catalysis in the Construction of Molecular Complexity
Abstract
Gold(I) complexes are the catalysts of choice for the cyclization of 1,6-enynes and related substrates under mild conditions.1 We have found that 1,n-enynes with alkoxy substituents at the propargylic position react with cationic gold catalysts by a new type of intramolecular 1,n-1-migration of OR groups.2 This reaction leads stereospecifically to tricyclic compounds related to the sesquiterpenes globulol, epiglobulol, and halichonadin F.
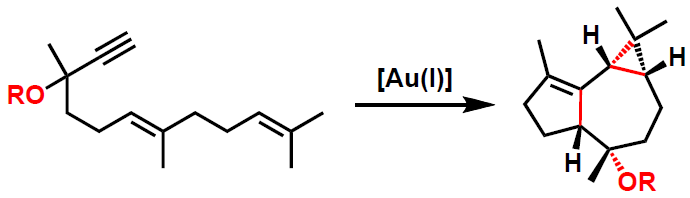
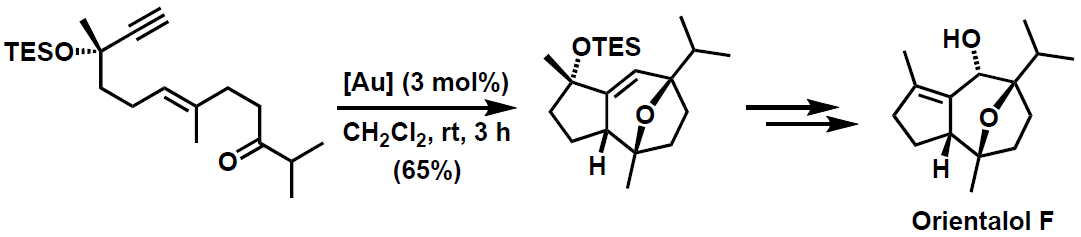
Similar substrates with carbonyl groups at the alkenyl chain react by a different pathway to form oxatricyclic compounds. This reaction has been applied for the synthesis of the sesquiterpene orientalol F.3
Related work on the synthesis of englerin A, a natural product that inhibits the growth of renal cancer cell lines at the nanomolar level,4 and recent fundamental work on the reactions of alkenes with alkynes with gold catalysts will also be presented.
References
- E. Jiménez-Núñez, A. M. Echavarren, Chem. Rev. 2008, 108, 3326-3350.
- E. Jiménez-Núñez, M. Raducan, T. Lauterbach, K. Molawi, C. R. Solorio, A. M. Echavarren, Angew. Chem. Int. Ed. 2008, 48, 6152-6155.
- E. Jiménez-Núñez, K. Molawi, A. M. Echavarren, Chem. Commun. 2009, 7327-7329.
- R. Ratnayake, D. Covell, T. T. Ransom, K. R. Gustafson, J. A. Beutler, Org. Lett. 2009, 11, 57-60.





