Novel potent inhibitors of key purine salvage enzymes
PI: Zlatko Janeba, co-PIs: Helena Mertlíková-Kaiserová, Pavlína Maloy Řezáčová
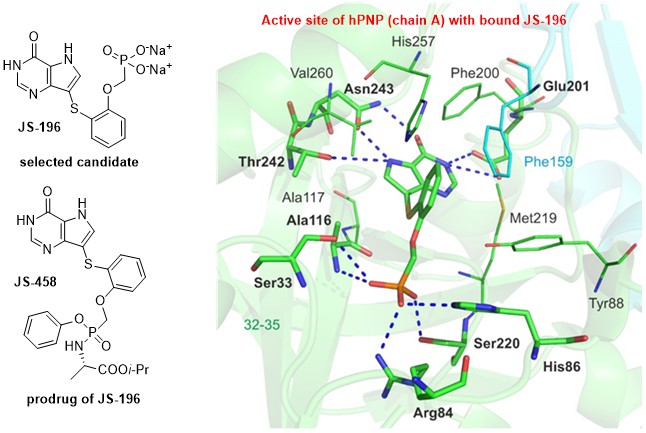
Purine nucleoside phosphorylase (PNP) represents a key enzyme of the purine salvage pathway and is highly clinically relevant as drug target (namely human (hPNP) and parasitic enzymes). Approval of forodesine (Japan, 2017) for treatment of peripheral T-cell lymphoma further validated hPNP as a druggable target with potential clinical applications. The main goal of this project was design and synthesis of potent hPNP inhibitors and subsequent selection of the best candidate for its preclinical evaluation as potential treatment of T-cell leukemias.
A large series of hPNP inhibitors was designed, synthesized, and many of them proved to be potent hPNP inhibitors, with IC50s as low as 2 nM. Many compounds also exhibited very potent and selective toxicity towards leukemic cell lines (CCRF-CEM, MOLT-4, Jurkat). Several PK and PD studies were performed with lead compound JS-196, as well as three in vivo proof-of-concept studies (chick embryos, mouse xenograft models). Certain therapeutic potential of compound JS-196 towards T-ALL leukemias was observed in all of the studies and the data confirmed that hPNP inhibition represents a viable treatment strategy. In future, problems with oral bioavailability of our candidate(s) will be addressed (formulation, prodrugs, etc.).
The research was performed as a multidisciplinary project based on the collaboration within IOCB, with Prof. Pospíšilová (CEITEC Brno) and Prof. Doubek (University Hospital Brno), and with our colleagues from Gilead Sciences.
PATENT:
- J. Skácel, Z. Janeba, H. Mertlíková Kaiserová: Inhibitors of purine nucleosidephosphorylase – synthesis and use thereof for treatment of T-cell acute lymphoblastic leukemia and lymphoma. WO 2021/083438 A1



