On the new character of complexes of fullerenes with piperidine: evidence of N→C dative/covalent bond
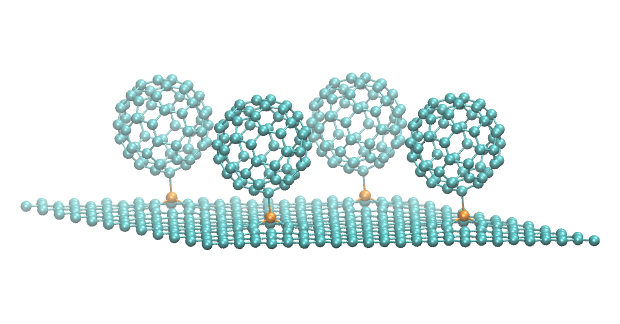
A team of researchers from the Pavel Hobza Group at IOCB Prague, Palacký University Olomouc (Czech Republic), and Luoyang Normal University (China) reported in Angewandte Chemie results of the combined experimental and computational studies on complexes formed between carbon allotropes (C20 and C60 fullerenes, graphene and single wall carbon nanotubes) and piperidine.
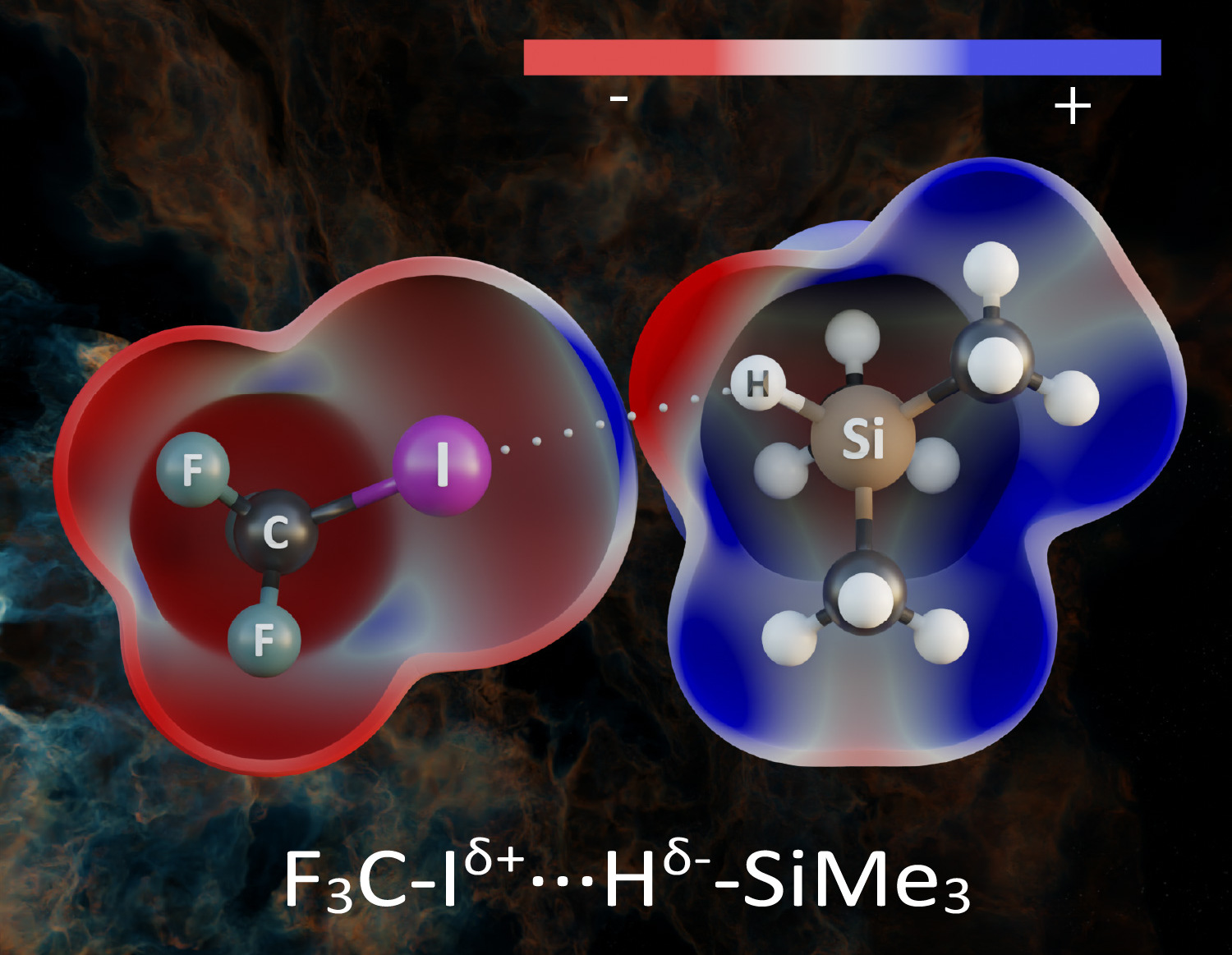
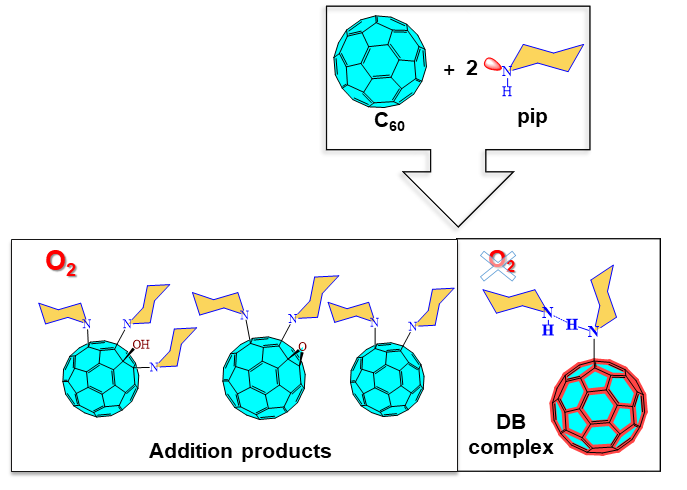
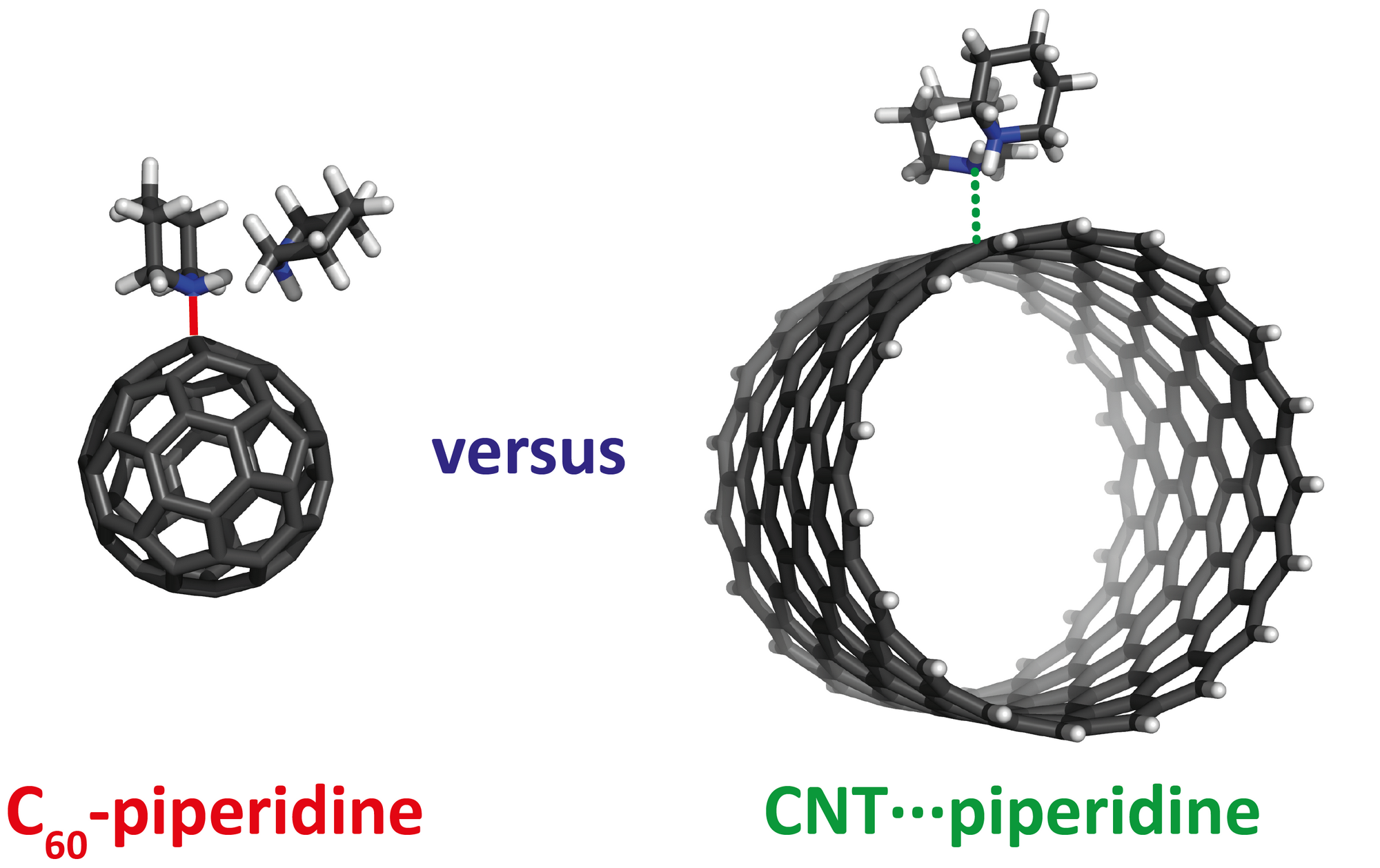
Buckmisterfullerene C60, a widely known ball-shaped compound, and its derivatives attract the researcher’s attention due to their applications in bio-related and material-chemistry fields. The studies proved the existence of the unexpected N→C dative/covalent bond, formed solely in complexes of fullerenes with piperidine. Non-planarity and five-member rings of carbon allotropes represent the essential structural prerequisites for the unique formation of a dative N→C bond. The thermodynamics calculations, molecular dynamics simulations, and NMR and FT-IR spectroscopies explain the specific interactions between C60 and piperidine.
The differences in the behavior of individual carbon allotropes in terms of dative bonding formation brings new insight into their controllable organic functionalization. These results provide information about the reactivity-structure relations of carbon allotropes that are potentially useful for their simple and selective covalent functionalization, applicable to a broad portfolio of nanotechnologies.
New findings on the character of interaction of piperidine and other secondary amines with C60 are important for the rational design of modified fullerenes and their applications in electrocatalysis, spintronics, and energy storage.
Original paper: Lamanec, M., Lo, R., Nachtigallová, D., Bakandritsos, A., Mohammadi, E., Dračínský, M., Zbořil, R., Hobza, P. and Wang, W. (2020), The Existence of a N→C Dative Bond in the C60–Piperidine Complex. Angew. Chem. Int. Ed. https://doi.org/10.1002/anie.202012851