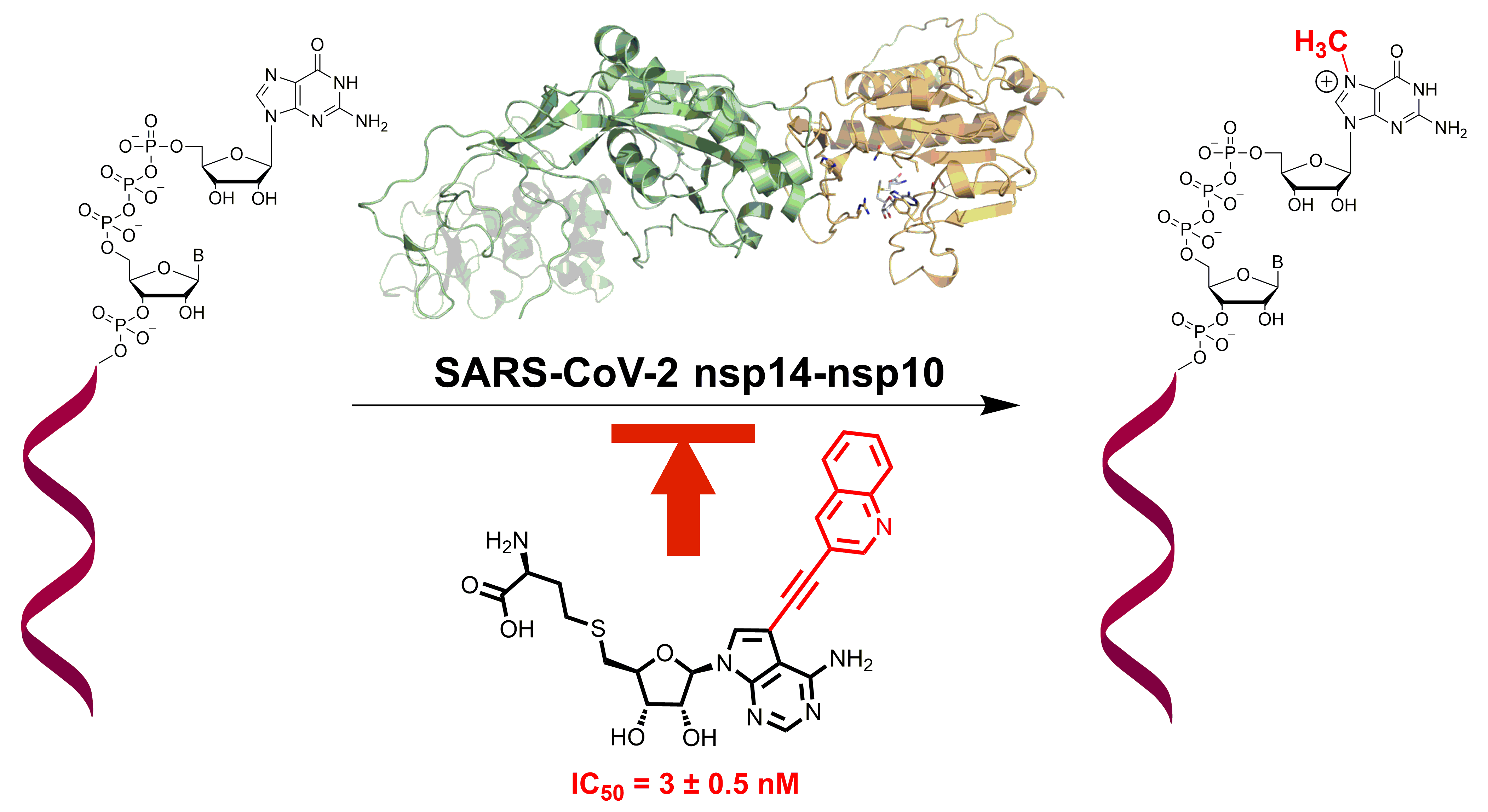
The group of Radim Nencka from IOCB Prague in collaboration with Evžen Bouřa and colleagues from the University of Toronto designed and synthesized novel small molecule-based inhibitors of the SARS-CoV-2 nonstructural protein nsp14.
The SARS-CoV-2 genome encodes for two methyltransferases (MTases). One of them is part of the nsp14 protein and is essential for mRNA stability, thus translation and viral spread in human cells.
As the crystal structure of SARS-CoV-2 nsp14 is unknown, the scientists designed the inhibitors using the solved crystal structure of highly homological SARS-CoV nsp14. The inhibitors are modified derivatives of the S-adenosyl-L-homocysteine (SAH), which is a side-product of the enzymatic methylation. The research has been published in ACS Infectious Diseases with Tomáš Otava as the first author.
Read the paper:
- Otava, T.; Šála, M.; Li, F.; Fanfrlík, J.; Devkota, K.; Perveen, S.; Chau, I.; Pakarian, P.; Hobza, P.; Vedadi, M.; Boura, E.; Nencka, R. The Structure-Based Design of SARS-CoV-2 nsp14 Methyltransferase Ligands Yields Nanomolar Inhibitors. ACS Infectious Diseases 2021. https://doi.org/10.1021/acsinfecdis.1c00131