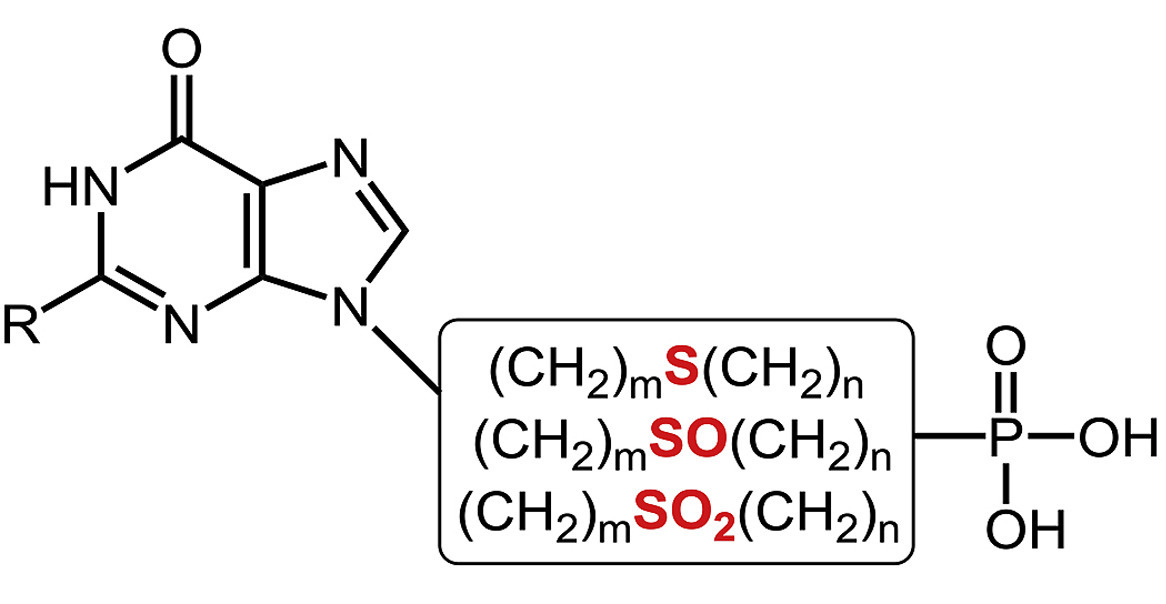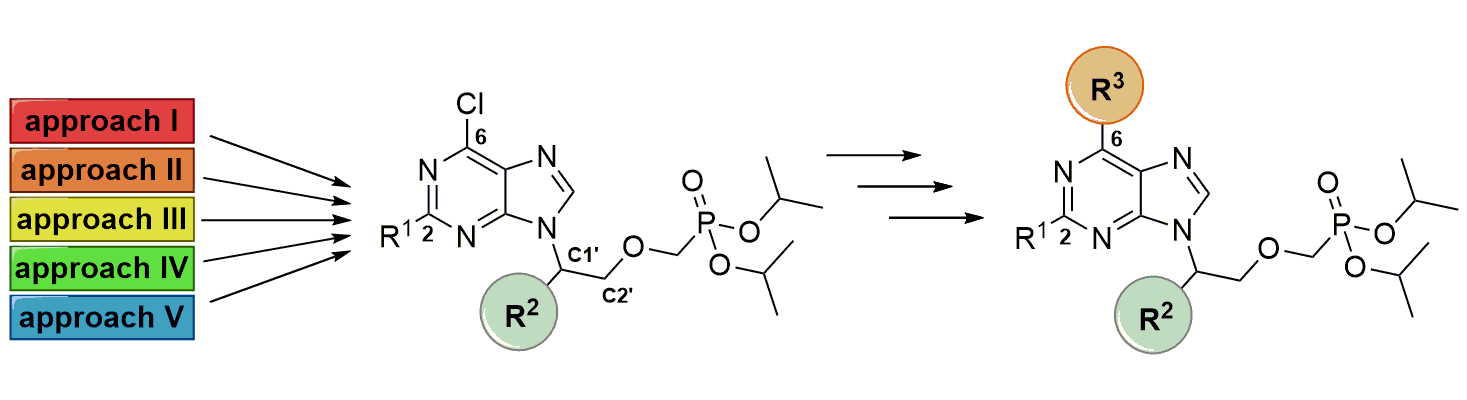
Filip Kalčic and Zlatko Janeba from IOCB Prague optimized some of the existing synthetic approaches leading toward C1'-branched acyclic nucleoside phosphonates (ANPs) and developed several new ones. ANPs are stable analogs of nucleotides and are well-known for their antiviral, anticancer, and antiprotozoal activity. Surprisingly, the synthesis and biological properties of C1'-branched ANPs have been so far rather overlooked.
These newly modified and optimized methods consist of shorter procedures giving higher overall yields. In addition, the final ANP derivatives bear 6-chloropurine moiety, which enables further modification of the nucleobase.
The paper is featured on the inside cover in Organic & Biomolecular Chemistry.

Read the paper
- Kalčic, F.; Dračínský, M.; Janeba, Z. Diverse synthetic approaches towards C1'-branched acyclic nucleoside phosphonates. Org. Biomol. Chem. 2021, 19, 6958. https://doi.org/10.1039/D1OB00751C