Experiment and theory in synergy: Understanding desaturation activity through rational enzyme mutagenesis
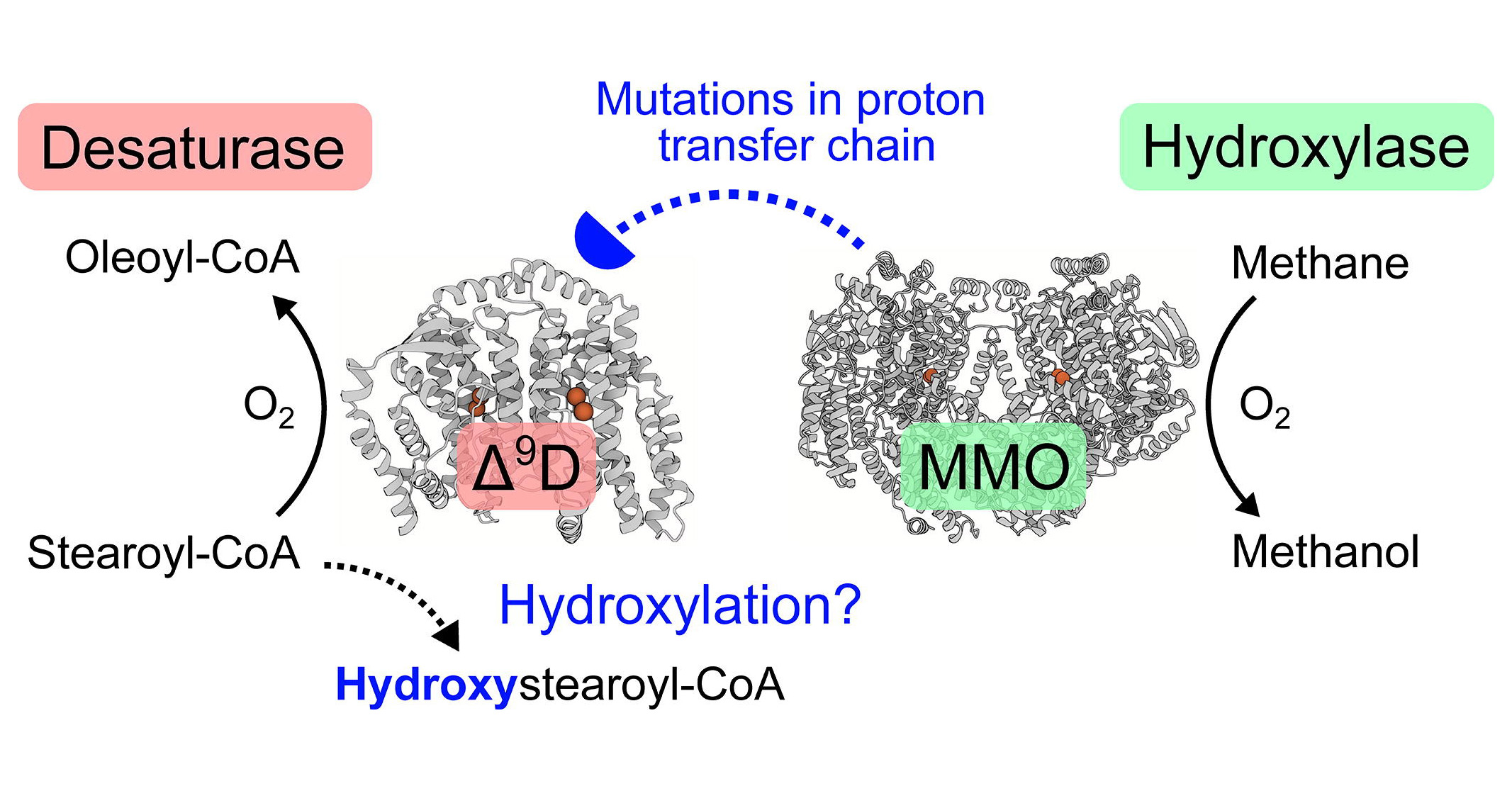
Mono- and binuclear non-heme iron enzymes catalyze a multitude of C–H bond functionalizations in alkyl chains and aromatic rings. One of the examples is the soluble chloroplastic enzyme Δ9 desaturase (Δ9D) involved in the unsaturated fatty acid biosynthesis in plants.
Researchers from Iva Pichová (Stanislav Macháček) and Lubomír Rulíšek groups (Michal Tupec, Martin Culka, and Daniel Bím), supported by the synthetic efforts of Aleš Machara, made use of the recently proposed reaction mechanism of soluble Δ9 desaturase (Δ9D) and identified auxiliary amino acid residues localized near the di-iron active site that is involved in the desaturation mechanism.
The scientists wanted to know if the perturbation of the auxiliary residues leads to the impairment of the desaturation reaction and a possible amplification of other minor reaction pathways, such as hydroxylation. For this reason, they made specific single-point mutations in Δ9D and determined the reaction products of mutants. The mutations promoted residual Δ9D monohydroxylation activities on stearoyl-CoA at the expense of native desaturation activity.
The results not only support the recently described reaction mechanism of the enzyme (Bím et al., JACS 2020, 142, 10412) but may open new horizons in a computationally aided redesign of the natural activity of enzymes.
Read the paper:
- Tupec, M.; Culka, M.; Machara, A.; Macháček, S.; Bím, D.; Svatoš, A.; Rulíšek, L.; Pichová, I. Understanding desaturation/hydroxylation activity of castor stearoyl Δ9-Desaturase through rational mutagenesis. Comput. Struct. Biotechnol. J. 2022, 20, 1378-1388. https://doi.org/10.1016/j.csbj.2022.03.010



