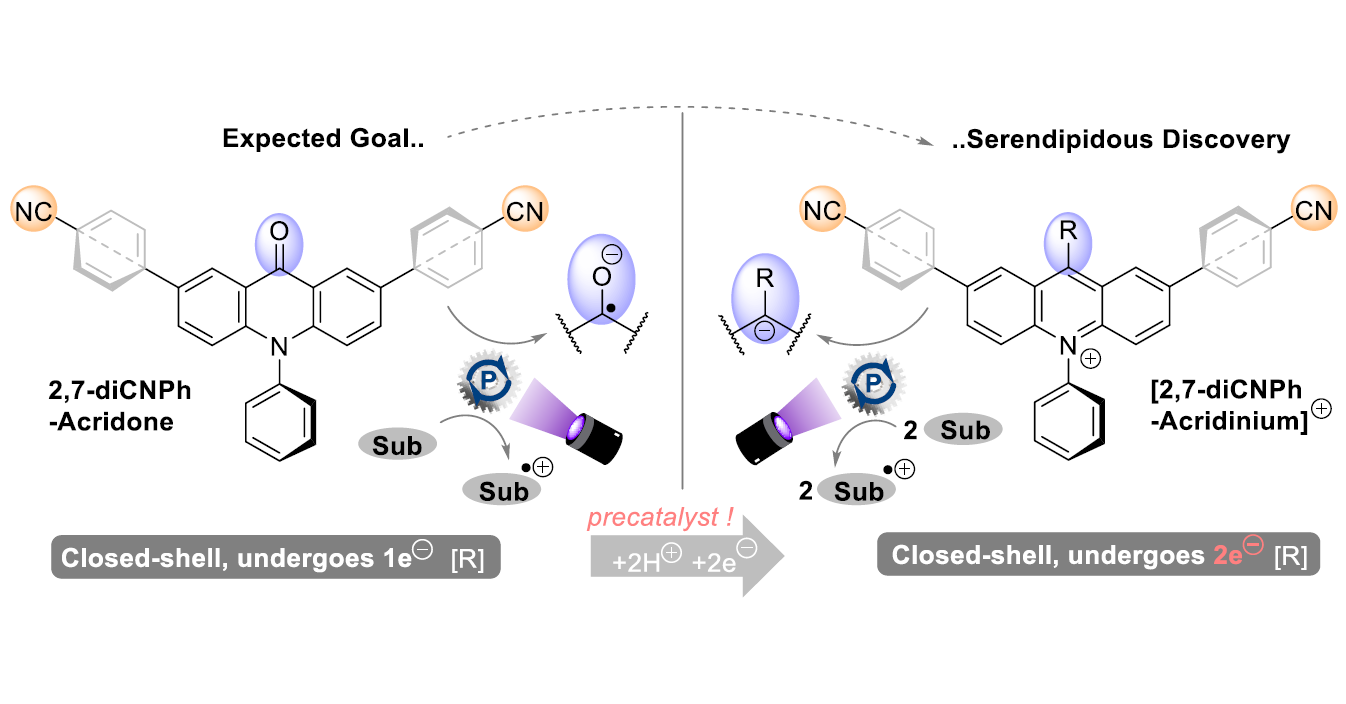
What if the “catalysts” chemists add to the reaction mixture are not the actual catalysts and only decompose to another catalytically active species? This question was asked by scientists from Tomáš Slanina Group at IOCB Prague in collaboration with Joshua P. Barham from the University of Regensburg while studying novel acridone catalysts as potent neutral single-electron-transfer (SET) photooxidants.
The researchers discovered that the acridone transformed under the reaction conditions into a novel acridinium salt with a record-breaking oxidation potential that appears to function by two-electron reduction of its excited state. This mechanism is very surprising as photoexcited acridinium salts have been reported only as one-electron oxidants until now.
Original article
- Žurauskas, J.; Boháčová, S.; Wu, S.; Butera, V.; Schmid S.; Fomański, M.; Slanina, T.; Barham, J. P. Electron-Poor Acridones and Acridiniums as Super Photooxidants in Molecular Photoelectrochemistry by Unusual Mechanisms. Angew. Chem. Int. Ed. 2023, e202307550. https://doi.org/10.1002/anie.202307550