Fibrillization blockers, a new tool in the fight against Parkinson’s disease
22 November 2019
Parkinson's disease is the second most common neurodegeneration. One of the key processes occurring during its development is the aggregation of the neuronal protein α-synuclein (αSyn). In its native state, this protein is disorganized and soluble, but it forms polymeric amyloid fibrils in the pathological tissue.
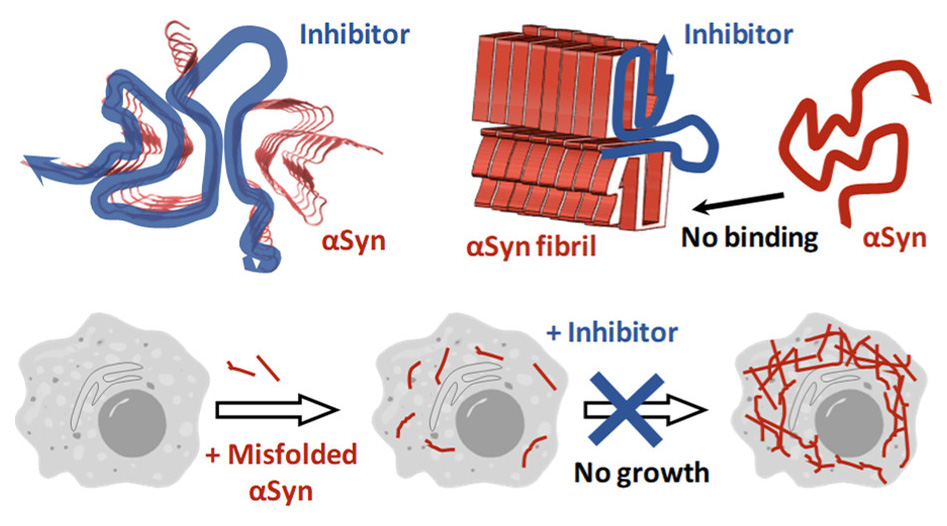
This fibrillization can be inhibited by molecules that bind to native αSyn, but in that case, the inhibitor concentration has to be close to total αSyn concentration in the cell.
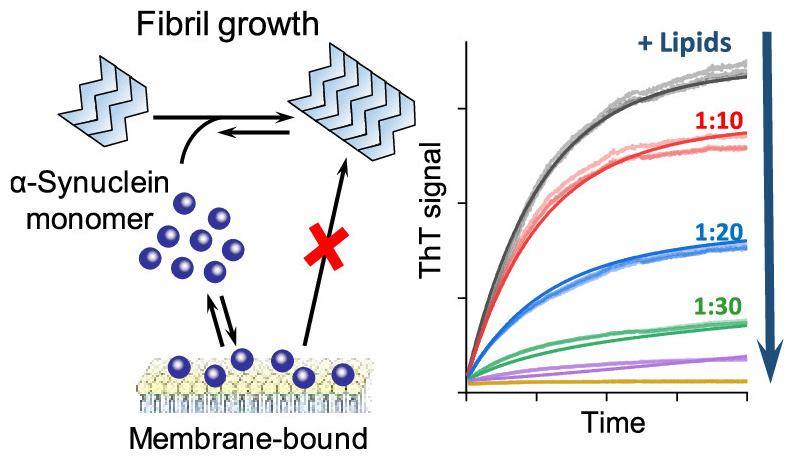
The team of scientists led by Volodymyr Shvadchak and Dmytro Yushchenko from IOCB Prague chose a different strategy. They targeted the growing amyloid fibrils, exploiting the fact that they grow only on their ends, by attaching the free αSyn and misfolding it to the amyloid form. Thus, blocking the ends of the fibrils and preventing them from further growth seemed like a natural choice. Also, the concentration needed for successful inhibition would then have to be comparable to the amount of fibril ends, not the total concentration of free αSyn.
After an attempt to block the ends with a conjugate of αSyn and a bulky globular protein, they successfully tried to enhance the affinity of the inhibitor to the fibril ends by dimerization, targeting the double-rod-like structure of the fibrils. The surprising finding was that even the αSyn derivative dimer without the bulky blocking group inhibited the fibril elongation.
This allowed the researchers to prepare several useful αSyn dimer derivatives differing in the linker that all exhibited high inhibitory capability against fibril elongation. Some seem to be stable in the cellular environment, making them a potential starting point for a novel approach to slow down Parkinson’s disease development, while others bear a fluorescent marker or a photocleavable linker, allowing for further studies of the fibrillization process. Especially the photocleavable one seems promising because it can be used to restart the fibril elongation at a defined time, helping further studies.
Original paper: Yevhenii A. Kyriukha, Kseniia Afitska, Andrii S. Kurochka, Shubhra Sachan, Maksym Galkin, Dmytro A. Yushchenko, and Volodymyr V. Shvadchak. α-Synuclein Dimers as Potent Inhibitors of Fibrillization. Journal of Medicinal Chemistry Article ASAP. DOI: 10.1021/acs.jmedchem.9b01400
Read next...