Researchers from IOCB Prague led by Iva Pichová have recently uncovered how an enzyme in Mycobacterium smegmatis, called GMP reductase (GMPR), helps maintain the balance of essential cellular metabolites known as purines. Purines, which include molecules such as ATP and GTP, have many crucial functions, from building DNA to powering fundamental cellular processes. This research identifies a novel shape-shifting mechanism in which GMPR changes its structure to turn itself on or off depending on the cell's immediate needs.
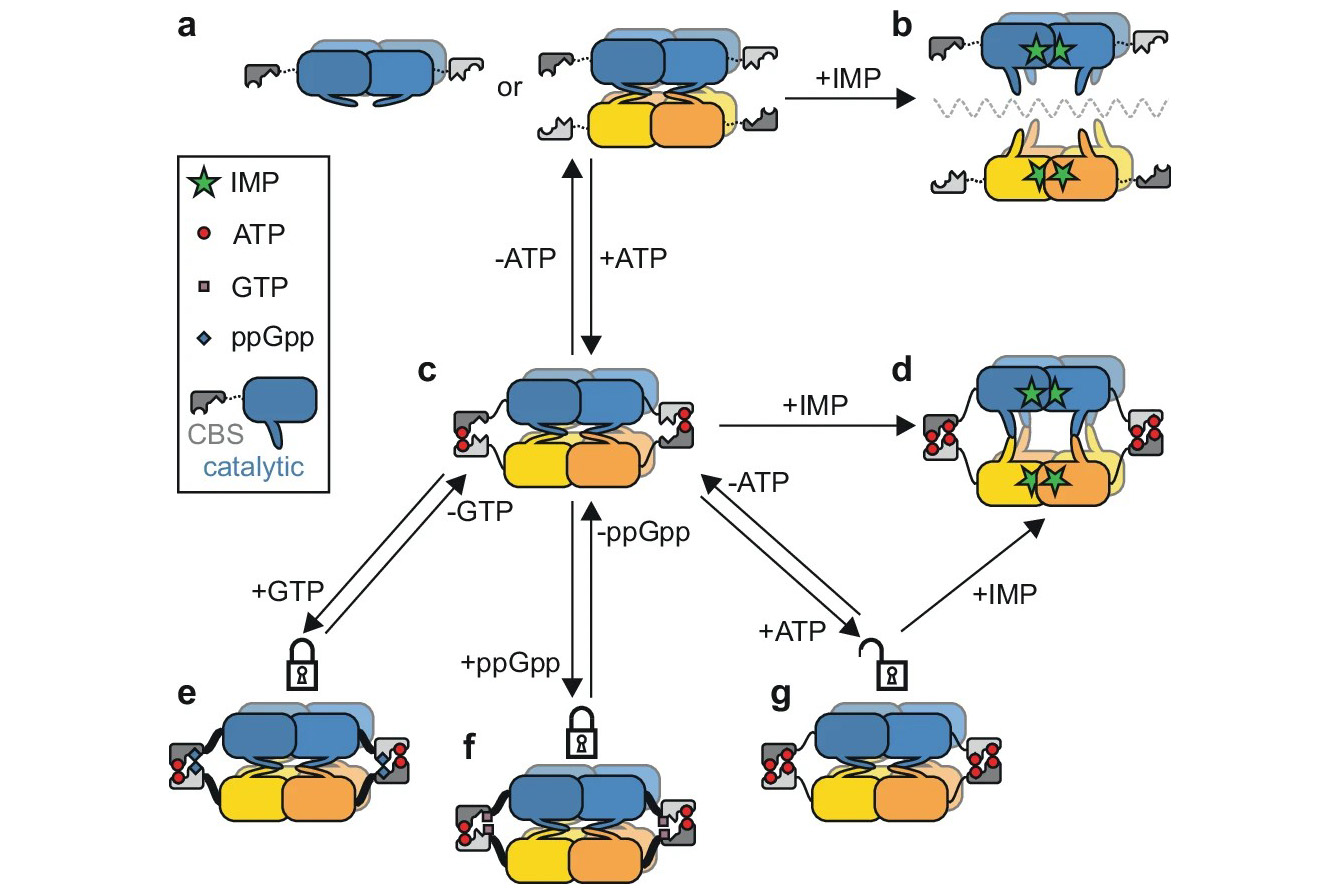
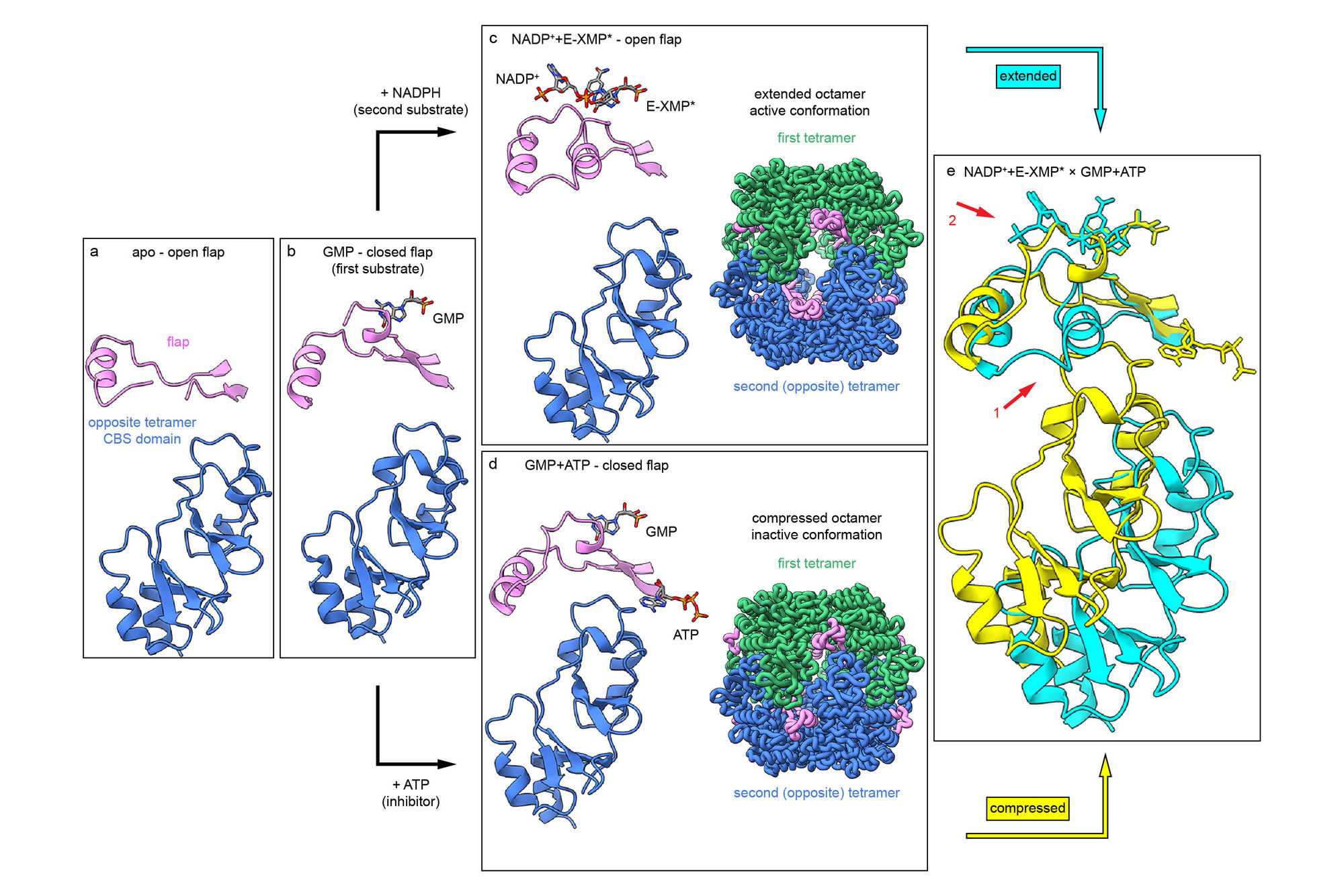
The researchers reveal that GMPR operates through a lock mechanism controlled by the competing signals of ATP and GTP. When ATP binds to the enzyme, it stabilizes a compressed conformation. This compression effectively blocks the enzyme from completing its chemical reaction. Conversely, GTP competes with ATP for the same binding site, preventing the enzyme from locking up and keeping it in an active state so it can continue processing nutrients.
This discovery uncovers a previously unknown way certain bacteria regulate their internal chemistry. While GMPRs in many organisms lack this built-in control switch, mycobacteria have evolved this specialized sensor to respond rapidly to changes in their chemical environment. The findings help explain how mycobacteria such as Mycobacterium smegmatis maintain homeostasis, the stable internal state necessary for life.
Read the paper
- Doležal, M.; Knejzlík, Z.; Kouba, T.; Filimoněnko, A.; Šváchová, H.; Dedola, M.; Klíma, M.; Pichová, I. Structural Basis for Allosteric Regulation of Mycobacterial Guanosine 5′-Monophosphate Reductase by ATP and GTP. Nat. Commun. 2026. https://doi.org/10.1038/s41467-026-71657-9