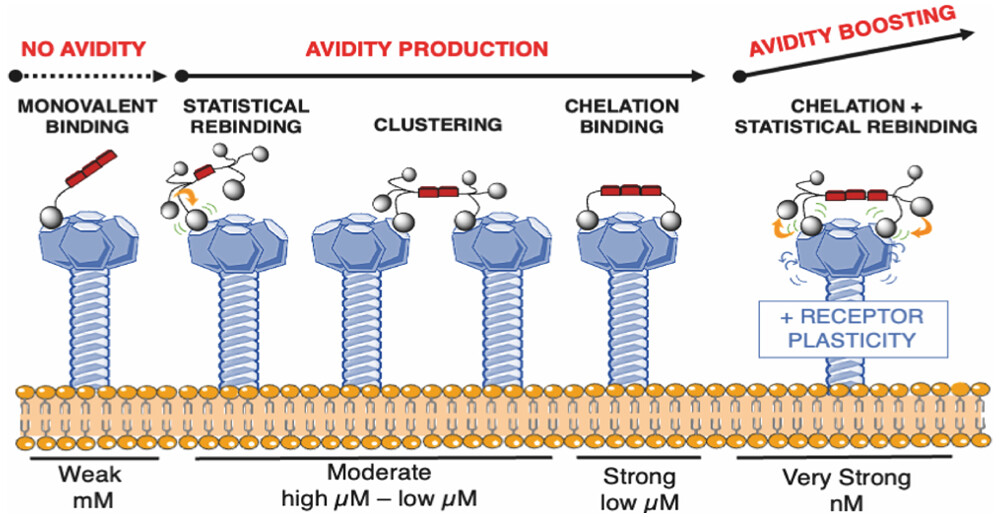
A new study from Martin Lepšík from IOCB Prague as one of the first authors provides a detailed understanding of interactions between multivalent virus-attachment blockers and DC-SIGN, a multimeric C-type lectin receptor. This receptor plays an important role in human immune responses to pathogens and has been highlighted as the co-receptor for the spike protein of the SARS-CoV-2 virus.
Researchers from IOCB Prague, the University Grenoble Alpes, the University of Milan, the University of Leeds, and the University of Bologna studied a series of dendrimer constructs with a rod core rationally designed to engage simultaneously two binding sites of the tetrameric DC-SIGN. Using molecular modeling, they were able to gain deeper insight into the role of different binding modes, which in combination produce a construct with a nanomolar affinity despite a limited valency.
The study published in ACS Central Science opens the way to the development of new virus attachment blockers adapted to different C-type lectin receptors of viruses.
Read the paper: Porkolab, V.; Lepšík, M.; Ordanini, S.; St John, A.; Le Roy, A.; Thépaut, M.; Paci, E.; Ebel, C. Bernardi, A.; Fieschi, F. Powerful Avidity with a Limited Valency for Virus-Attachment Blockers on DC-SIGN: Combining Chelation and Statistical Rebinding with Structural Plasticity of the Receptor. ACS Cent. Sci. 2023. https://doi.org/10.1021/acscentsci.2c01136