Cryo-EM structures show how Trypanosomes escape the human innate immune response
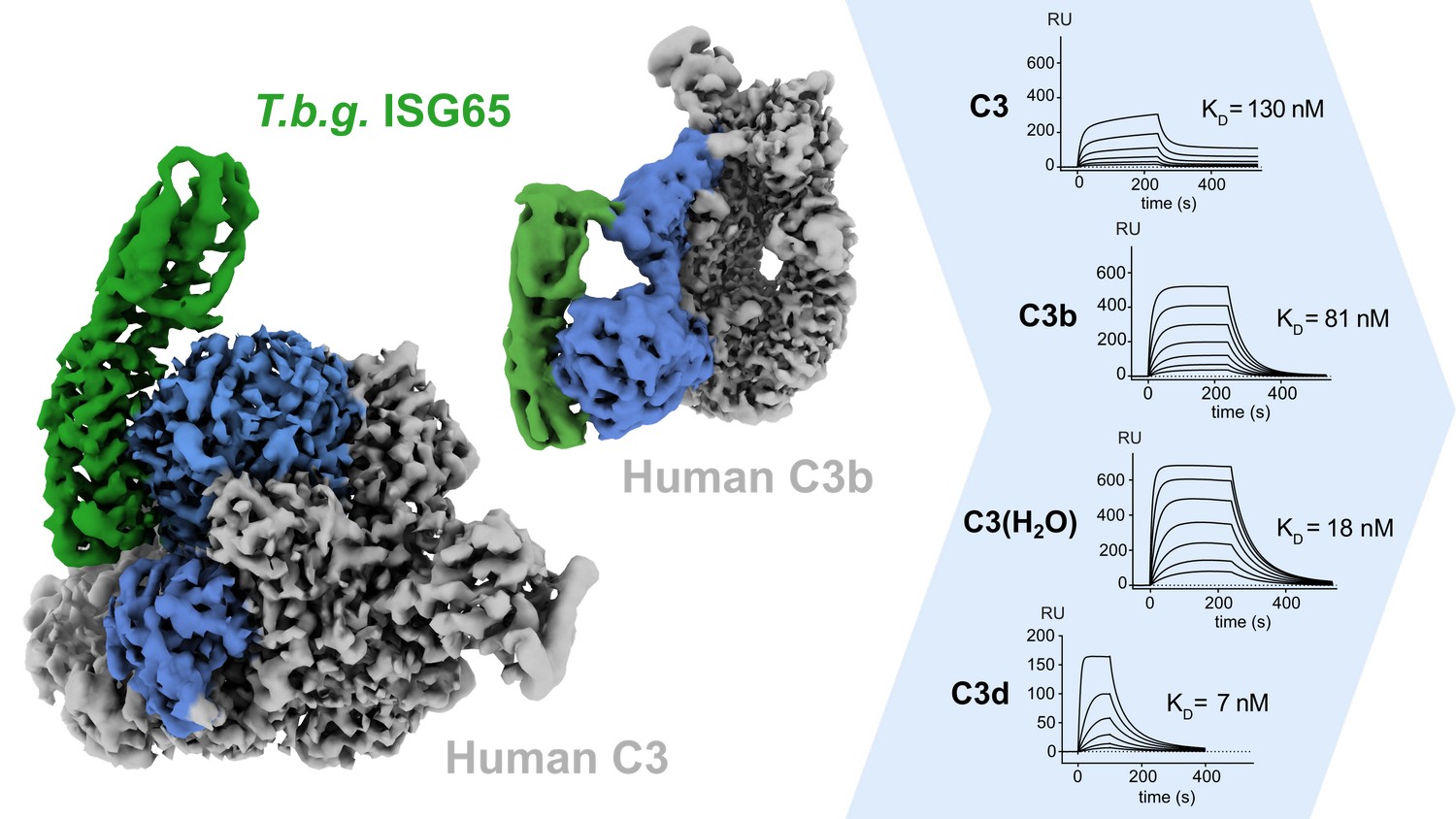
Researchers from Sebastian Zoll’s group at IOCB Prague determined the first cryo-EM structures of a surface receptor of the human-infective parasite Trypanosoma brucei gambiense in complex with human complement factor C3 in different conformations. This provides the structural basis to understand how the parasite escapes the immune response at an early stage of infection.
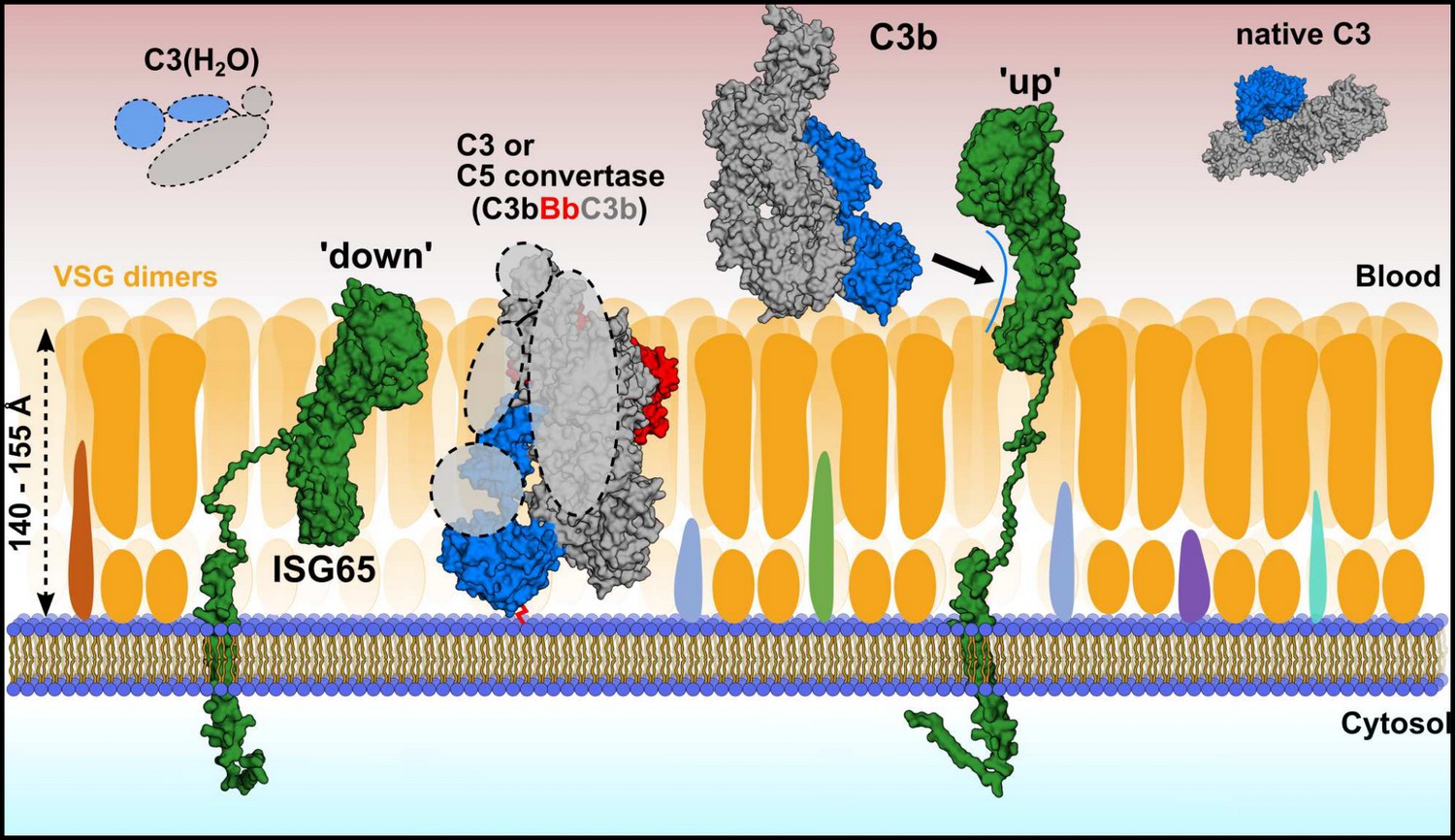
While most pathogens have found ways to avoid the deleterious effects of the immune system by escaping neutralizing antibodies and other effector proteins inside cells or less accessible tissues, African trypanosomes are hiding in plain sight and proliferate within the human blood stream. The parasites are exposed to the full pressure of both branches of the immune system and must employ a plethora of countermeasures to avoid clearance from the cardio-vascular system.
The mechanisms of this ‘molecular warfare’ have been identified and, particularly with regard to the adaptive immune response, described in detail. However, our understanding of innate immune defence mechanisms had remained incomplete until now.
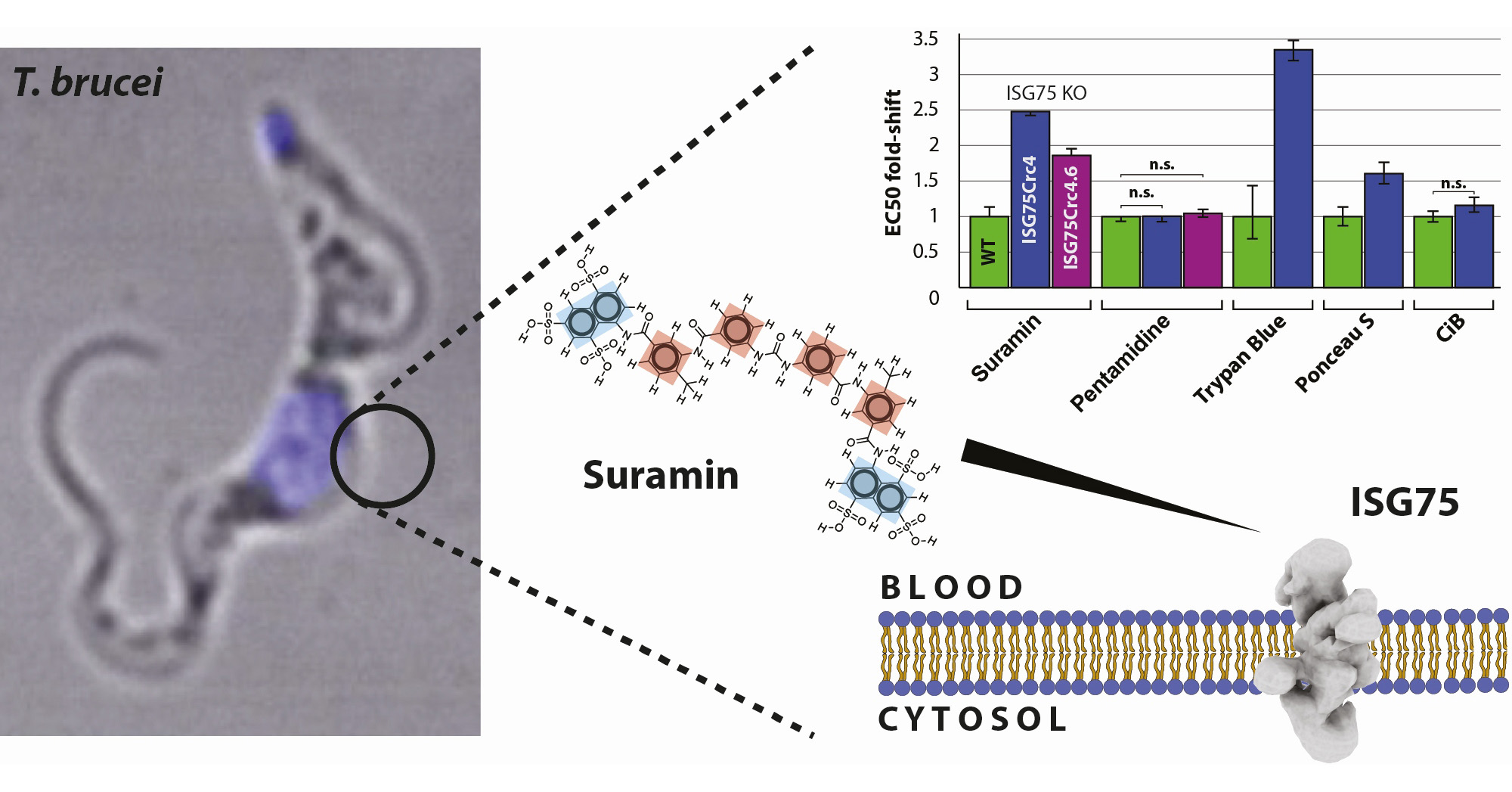
Under the leadership of Sebastian Zoll, a team of researchers, with Hagen Sülzen as the first author of the study, and including Petr Pompach and Martin Zoltner from BIOCEV, employed an integrative structural biology approach and demonstrated that the interaction between ISG65 and C3, the central hub of the human complement system, is inhibitory and remarkably specific. Inhibiting only a single pathway within the complement system allows the pathogens to maintain a tailored and efficient immune defence.
The study was published in Nature Communications and shows the first 2 cryo-EM structures from IOCB Prague alongside one of the first cryo-EM – Alphafold hybrid models that was deposited to the PDBdev.
The original article:
- Sülzen, H., Began, J., Dhillon, A. et al. Cryo-EM structures of Trypanosoma brucei gambiense ISG65 with human complement C3 and C3b and their roles in alternative pathway restriction. Nat Commun 14, 2403 (2023). https://doi.org/10.1038/s41467-023-37988-7